Physics Questions and Answers
If you want to learn more about the nature and properties of matter and energy or you're simply preparing for a Physics exam, these Physics past questions and answers are ideal for you.

If you want to learn more about the nature and properties of matter and energy or you're simply preparing for a Physics exam, these Physics past questions and answers are ideal for you.
Specific heat capacity of ice = 2100 \(Jkg^-1\) \(k^-1\)
6680 J
1680 J
7520 J
7940 J
Correct answer is D
Quantity of heat required to raise the temperature of the ice cube from \(-10^oC\) to \(0^oC\)
⇒H=mc►ø = 0.02 × 2100 × 0 -(-10)
⇒ H = 0.02 × 2100 × (0 + 10) = 0.02 × 2100 × 10
⇒ H = 420 J
quantity of heat required to melt ice at \(0^oC\)
⇒ H = mL = 0.02 × 3.34 × \(10^5\)
⇒H=6680 j
Quantity of heat required to raise the temperature of the melted ice cube (water) from \(0^oC\) to \(-10^oC\)
⇒H=mc►ø = 0.02 × 4200 × (10-0)
⇒H = 0.02 × 4200 × 10
⇒H = 840j
;420 + 6680 + 840 = 7940 j
3.54nC
42.5nC
35.4nC
4.25nC
Correct answer is B
A= \(0.8m^2\) d= 20mm =\(\frac{20}{1000}\) = 0.02m
v =120v; \(ε_oA\)= \(8.85 × 10^-12 fm^-1\)
C = \(\frac{ε_oA}{d}\)
C =\(\frac{8.85 × 10^-12×0.8}{0.02}\)
C= \(3.54 × 10^-10 F\)
Q= CV
⇒ \(3.54 × 10^-10\) × 120 = \(4.25×10^-8c\)
Q=\(42.5 × 10^-9c\) = 42.5nC
6.43 cm
8.24 cm
4.26 cm
5.82 cm
Correct answer is D
\(r_1\) = 4.5cm , \( P_1\) =is the total pressure on the bubble at a depth of 12m from the surface.
\(P_1\) = 12 + 10.34 =22.34m
\(V_1\) = \(\frac{4}{3}\)π× \(r^3_1\)
= \(\frac{4}{3}× π×{4.5^3cm^3}\)
\(P_2\) = 10.34m
\(V_2\) = \(\frac{4}{3} {π}{r^3_2}\)
from boyles law:
\(P_1V_1\) = \(P_2V_2\)
⇒ 22.34× \(\frac{4}{3}× π×{4.5^3}\) = 10.34 × \(\frac{4}{3}×π×{r^3_2}\)
⇒ 22.34 × \(4.5^3\) = 10.34 × \(r^3_2\)
⇒ \(r^3_2 = \sqrt[3]{196.88}\)
⇒ \(r_2\) = 5.82cm
6.43 cm
8.24 cm
4.26 cm
5.82 cm
Correct answer is D
\(r_1\) = 4.5cm , \( P_1\) =is the total pressure on the bubble at a depth of 12m from the surface.
\(P_1\) = 12 + 10.34 =22.34m
\(V_1\) = \(\frac{4}{3}\)π× \(r^3_1\)
= \(\frac{4}{3}× π×{4.5^3cm^3}\)
\(P_2\) = 10.34m
\(V_2\) = \(\frac{4}{3} {π}{r^3_2}\)
from boyles law:
\(P_1V_1\) = \(P_2V_2\)
⇒ 22.34× \(\frac{4}{3}× π×{4.5^3}\) = 10.34 × \(\frac{4}{3}×π×{r^3_2}\)
⇒ 22.34 × \(4.5^3\) = 10.34 × \(r^3_2\)
⇒ \(r^3_2 = \sqrt[3]{196.88}\)
⇒ \(r_2\) = 5.82cm
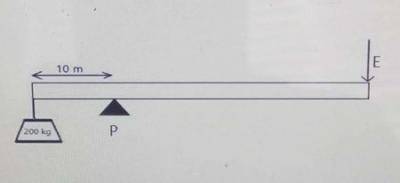
250
300
450
200
Correct answer is A
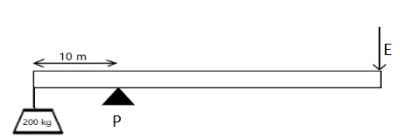
ε= 80%, L=200× 10 = 2000N, \(d_L\)=10m
\(d_E\)= 110-10 = 100m, E=?
ε = \(\frac{work done on the load}{work done by the effort}\) × 100%
⇒ work done on load = 2000 ×10 =20,000 j
⇒ work done by effort = E × 100 = 100E
⇒ 80 = \(\frac{20,000}{100E}\) × 100%
⇒ 80 = \(\frac{20,000}{E}\)
⇒ E = \(\frac{20,000}{80}\)
⇒ E = 250N
\(2.4×10^{10}(Nm^{-2})\)
\(1.5×10^{10}(Nm^{-2})\)
\(2.4×10^9(Nm^{-2})\)
\(1.3×10^{10}(Nm^{-2})\)
Correct answer is A
r=0.2mm = \(2 × {10^{-4}}\) e= 0.05% of L,=0.005L, m=1.5kgg=\(10 ms^{-2}\) , y=?,
(1000 mm = 1m)
Y = \(\frac{FL}{Ae}\)
w = mg = 1.5 × 10 = 15
A = \(\pi r^2\) = \(\frac{22}{7}×(2×10^{-4})^2\) = \(1.26×10^{-7}(m^2)\)
⇒ Y = \(\frac{15L}{1.26×10^{-7}×0.005L}\)
⇒ Y = \(\frac{15}{1.26×10^{-7}×0.005}\)
⇒ Y = \(\frac{15}{6.29×10^{-10}}\)
Y = \(2.4×10^{10}(Nm^{-2})\)
How much net work is required to accelerate a 1200 kg car from 10\(ms^{-1}\) to 15\(ms^{-1}\)
1.95×\(10^5 j\)
1.35×\(10^4 j\)
7.5×\(10^4 j\)
6.0×\(10^4 j\)
Correct answer is C
m=1200kg, \(V_1\)= \(10ms^{-1}\) \(V_2\) = \(15ms^{-1}\), w= ?
work=►K.E = \( K.E_2\) = \(K.E_2\) - \(K.E_1\)
⇒work= \(\frac{1}{2}{mv^2_2}-\frac{1}{2}{mv^2_1}\)
⇒work= \(\frac{1}{2}m({v^2_2}-{v^2_1}\))
⇒work= \(\frac{1}{2}× 1200× (15^2-10^2)\)
⇒work = 600 × (225 -100)
⇒work= 600 × 125
⇒work= 7.5×\(10^4 j\)
Which process is responsible for production of energy in stars?
Nuclear reaction
Nuclear fission
Nuclear fusion
Radioactive decay
Correct answer is C
Nuclear fusion is the process in which lighter nuclei are combined to form heavier ones. Matter lost in this process is converted into energy. The energy produced inside the stars is due to the process of nuclear fusion.
2.5kw
250kw
300kw
3.0kw
Correct answer is A
L=1.5m, A=\(620m^2\), ø=25-15=10°c
K=0.6071 , \(Wm^{-1}k^{-1}\) , \(\frac{q}{t}\) =?
\(\frac{q}{t}\) =KA, \(\frac{►t}{L}\)
where k is the thermal conductivity constant and \(\frac{q}{t}\) is the rate of heat transfer
=\(\frac{q}{t}\)=0.6071×620×\(\frac{10}{1.5}\)
\(\frac{q}{t}\) = 2509.35w ≈ 2.5kw
Information and Communication Manager at Nigerian Breweries Plc
Area Sales Manager at Airtel Nigeria
State Business Manager at Airtel Nigeria
Regional Technical Officer at Airtel Nigeria
Business Manager at Airtel Nigeria
Contract Administrator at Robert Walters
Import & Export Manager at Kerildbert Holdings Limited
JV Account Payable Officer at WTS Energy
HR Intern, Learning and Development at MainOne Cable
Senior Technical Officer - Endemic Diseases & NTDs (AfCDC) at the African Union